In this entry, we’ll learn about vaccination (through which phases a vaccine goes before it is approved and distributed) and how vaccine hesitancy can be tackled.
The first topic has already been discussed on an article of mine (science news), therefore it is here an iteration of the section.

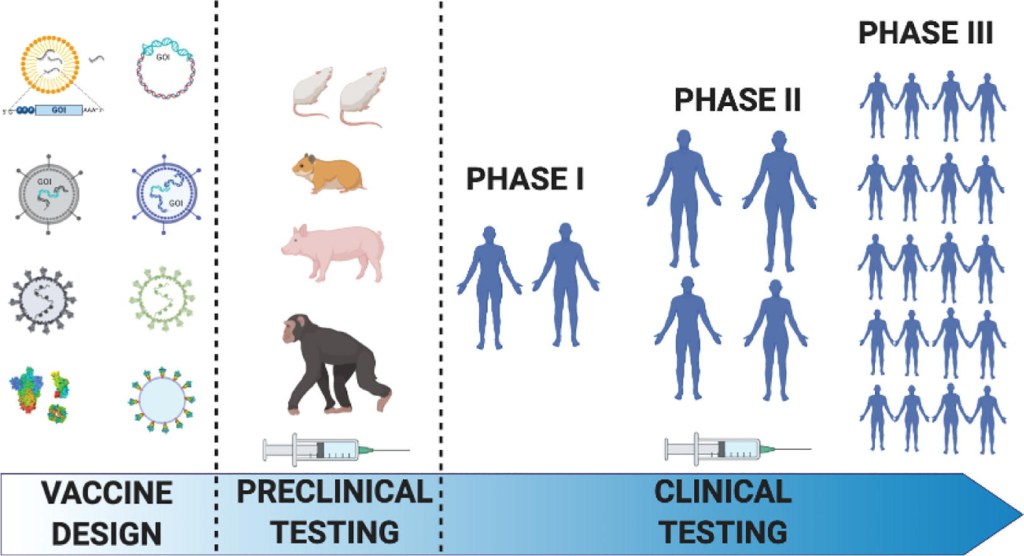
Vaccine Clinical Trial Phases
As I did in Science News (21st June, 2021), I’ll start with a very short summary that is based on the ScienceDaily summaries.
In order to ensure safety and efficacy of a new vaccine, clinical trials are conducted. Before a clinical trial is conducted, however, a pre-clinical trial must be completed.
Pre-clinical:
Before human trials; safety and effectiveness are tested and it is overseen by an independent ethics committee.
Phase 1:
Small trials, around 100 volunteers; safety is the main objective, immune response test.
Phase 2:
1,000 to 2,000 volunteers; still mostly safety, but also effectiveness included.
Phase 3:
Final trial before a vaccine is approved, about 10,000 volunteers; primarily focus on efficacy and whether the vaccine actually lowers infection.
Phase 4:
Data is still being collected on its safety and how well it works.
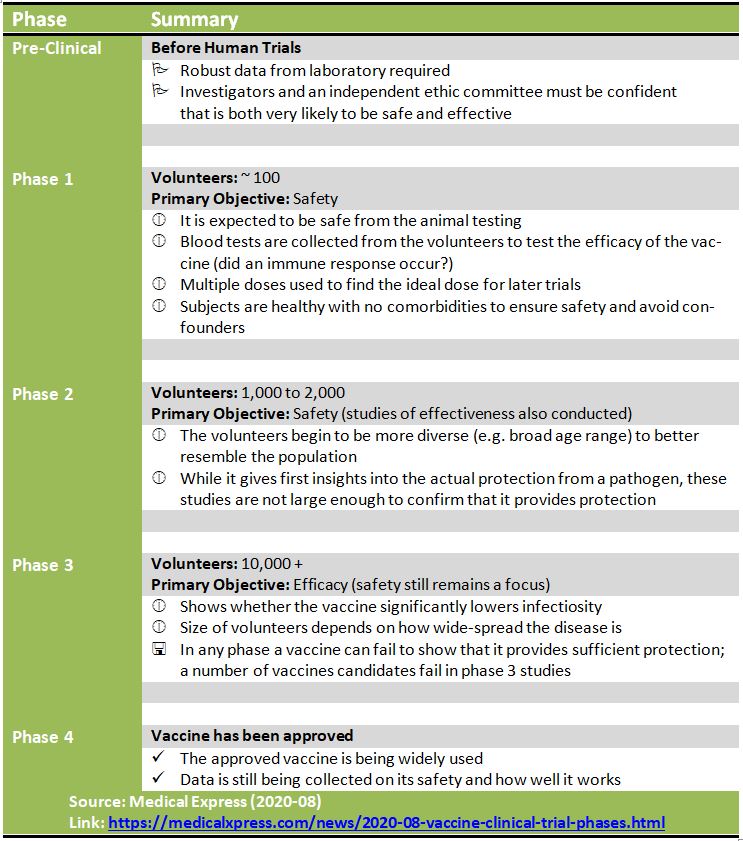
Now to the more elaborated summary (in tabular form):
The article of Medical Xpress also explains the rather fast pace of the vaccine development: „In the case of COVID-19, we have been fortunate to see sufficient investment that has enabled planning for all phases of clinical trials to take place at the beginning, allowing the next phase of clinical trials to commence quickly, so as soon as the data from the previous phase supports it. This has meant that we are seeing vaccine development happen at a rate much faster than ever before without compromising at all on any of the usual processes required to be certain about the vaccine’s safety, as well as how well it is likely to work.“
A lot of resources were shifted to the search for a vaccine and new programs brought into being such as HHS’ Operation Warp Speed (OWS), NIH’s Accelerating COVID-19 Therapeutic Interventions and Vaccines (ACTIV) partnership, NIH’s Rapid Acceleration of Diagnostics (RADx) initiative, and the WHO’s Solidarity Vaccine Trial (from the ScienceDirect study: „SARS-CoV-2 vaccines in advanced clinical trials: Where do we stand?“).
Source
https://medicalxpress.com/news/2020-08-vaccine-clinical-trial-phases.html
Study: Covid-19 vaccine acceptance and hesitancy in
low- and middle-income countries
Authors: Julio S. Solís Arce, Shana S. Warren, Niccolò F. Meriggi, et al.
Source: Nature Medicine
Published: 16th July, 2021
Link: https://www.nature.com/articles/s41591-021-01454-y
Method of Research
This study covers a large geographic across Africa, Asia and Latin America.
Overall, a total of 44,260 individuals were surveyed.
The measures used in this study have also been employed by other Covid-19 vaccine acceptance studies and follow the recommendations of the WHO Data of Action guidance to allow for meaningful cross-study and cross-country comparisons.
Summary of the Study
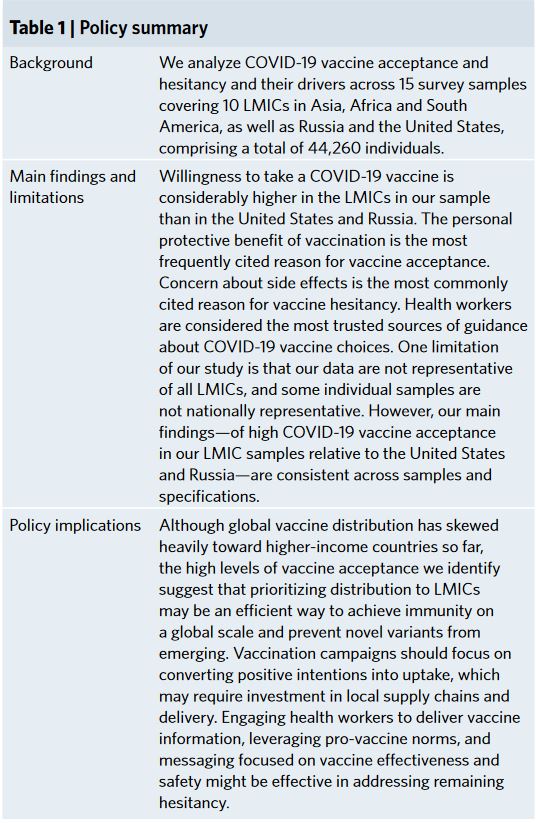
A summary was made by the authors as well, also included here (page 2):
Detailed Findings
Internal research within the Innovations for Poverty Action (IPA), the International Growth Center (IGC) and the Berlin Social Science Center (WZB) was conducted to select studies which are included in their sample.
Given the context of the pandemic, a survey modality (phone) was used that is both safe and appropriate for contexts with limited internet coverage.
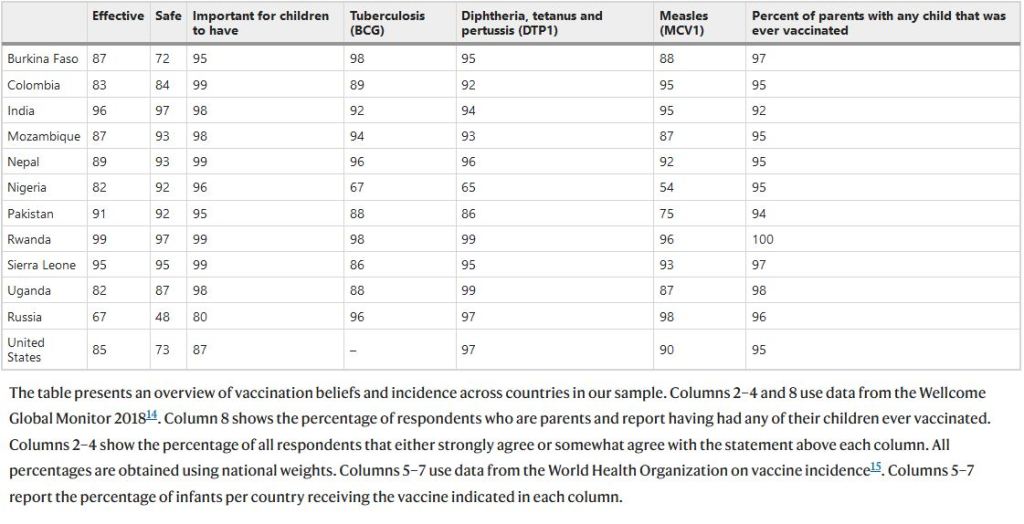
Vaccine Acceptance
- The acceptance rate is 80.3% across all LMIC studies (78.0% median)
- Pakistan and Burkina Faso are ranked the lowest
In Pakistan, the low acceptance rate may be linked to negative historical experiences
with foreign-led vaccination campaigns.
In Burkina Faso, the general vaccine hesitancy might be reflected. As it can be seen in
Table 2, fewer people believe that vaccines in general are safe (72%) than any other country except Russia (48%). - Women are generally less willing to accept vaccine than men
- Mixed evidence when it comes to the relationship between age and Covid-19 vaccine acceptance:
-> In India and Nigeria, for instance, respondents younger than 25 are less willing to take the vaccine while in Mozambique, Pakistan and Rwanda those under the age of 25 are „significantly more willing“ (p. 3)
Reasons given for Vaccine Acceptance
- Personal protection against Covid-19 (91%)
In the US 94% and Russia 76% – overall the most cited reason - Family protection is placed in the distant second place (average of 36%)
Reasons for Vaccine Hesitancy
- Concern about side effects (most frequently expressed)
In Sub-Saharan Africa it is the strongest: Uganda (81.5%), Sierra Leone (57.9%)
In Russia that concern is also present (36.8%) and more so in the US (79.3%) - A lack of concern regarding Covid-19 infection is another factor:
In the United States quite common (39.3%), as well as Pakistan (29.4%)
and Nepal (20.4%)
Most Trusted Source of Guidance (Covid-19 Vaccine)
- The health system is seen as the most trustworthy source (48.1% on average)
An exception is Rwanda (see page 7, Figure 3) - Health workers are cited most often in Sierra Leone (89.3%), Nigeria (58%) and
Burkina Faso (51.6%)
-> The trust in Sierra Leone’s health workers and Health Ministry may be reflected by the investments made in public health following the 2014-2015 Ebola epidemic - Countries that cited family and friends as the most trusted source are Colombia (36.6%), Nepal (35.6%), Russia (28.1%) and Burkina Faso (18.4%)
- Neither religious leaders nor celebrities are seen as top sources of guidance
-> Exception being Nepal where 16.1% trust famous people
Other
- In Rwanda, 34% of the respondents would trust ‚themselves‘ the most for guidance
- In the US, Joe Biden was cited was cited with 14% as the most trusted source (due to his position as president-elect, he was excluded from the government category)
Persuasion Campaigns – Approaches proposed by the Authors
In order for herd immunity to be achieved, vaccination must be administered to a large majority of the population. While policymakers and stakeholders must tailor their strategy specifically to the needs and problems of their respective country, three implications are highlighted.
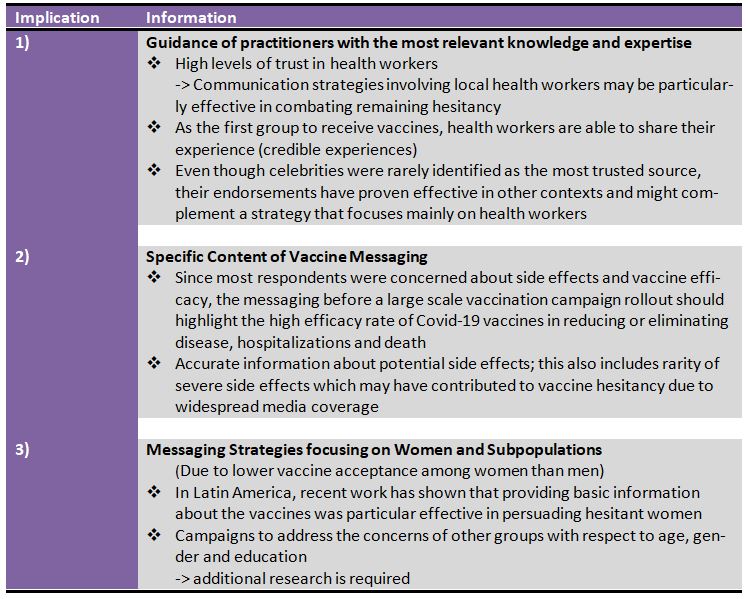
Considering the general pro-vaccination stance of many LMIC citizens, the availability of vaccines can increase the uptake considerably. In Colombia and Nepal, where family and friends are viewed as an important source of avdice, this might yield particularly strong.
The general acceptance of vaccines will be of key importance in the future, both for keeping old diseases in the graveyard (such as smallpox, eradicated in 1980 after a 20-year vaccination campaign of the WHO) and mitigating the effects of current diseases.
Another example of an effective vaccination campaign is polio which mostly affects children at the age of 3-8 years. The virus spreads from the lympth vessel and bloodstream and infects the nerve cells. Since children are still in their growth pase, the growth disturbances can lead to paralysis and damages to joints. Since 1988, the amount dropped from 350,000 to 33 in 2018 – a decline of over 99%.
I therefore end this blog entry with a section at the end I find important:
„Social signaling of positive attitude towards vaccines may help shift social norms toward even greater immunization acceptance and uptake in the community at large. As with messaging, policymakers should consider designing and evaluating social mobilization strategies targeted toward more hesitant subgroups.“ (p. 8)
Note: I still recommend reading the study, since I couldn’t include all the details in the summary.

